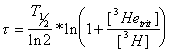 (1)
(1)
1. General background
Tritium (3H or T) is the radioactive isotope of hydrogen that decays with a half life of 12.43 years to its stable daughter 3He. Tritium is produced naturally in the upper atmosphere by interaction of nitrogen, and, to a lesser extent, oxygen with cosmic rays. After oxidation to HTO, it takes part in the natural water cycle. Before the full potential of natural tritium as a tracer for water movement in natural systems could be explored its distribution was masked by addition of large amounts of so-called ‘bomb tritium’ produced during the surface tests of nuclear weapons. These tests which were mainly performed in the early 1960s, led to an increase of tritium in precipitation over the continents of the northern hemisphere from roughly 5 TU to levels of the order of 1000 TU. One TU (Tritium Unit) means a tritium to hydrogen ratio of 10-18.
Whereas the addition of bomb tritium to the environment practically eliminated the use of natural tritium as a tracer, it offered a new tool, i.e., the use of the bomb tritium peak (Fig. 1) as a ‘dye’ that is delivered to natural water systems from the atmosphere on local to global scales. If the tritium delivery as a function of time can be reconstructed, this penetration process can be used for quantitative studies of water movement through identification of the bomb peak in certain ground water bodies. However, there are natural limits to this method because tritium decay and dispersion make it increasingly difficult to identify the bomb peak in groundwater.
These problems can be overcome by using tritium in combination with its decay product 3He (3Hetrit) as first suggested by Tolstykhin and Kamensky and experimentally confirmed by Torgersen et al. Simultaneous measurement of tritium and tritiogenic 3He allow us (1) to identify the tritium peak as the sum of tritium and (3Hetrit), [3H + 3Hetrit], even if most of the tritium has decayed, and (2) to directly calculate an age from the radioactive mother/daughter ratio (tritium/3He age). Extensive studies of the methodology and applicability of tritium/3He dating were started in the mid/late 1980s and early 1990s.
2. Determination of the tritium/3He age
In principal, the determination of the tritium/3He age of groundwater is simple. If both the tritium and 3Hetrit concentrations are measured in TU, it can be calculated as
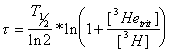 (1)
(1)
The most difficult step in the determination of the tritium/3He age is the separation of 3Hetrit from the total 3He (3Hetot) dissolved in a groundwater sample. The total 3He concentration has a variety of sources (equation (2)):
![]() (2)
(2)
where 3Heeq = 3He in solubility
equilibrium
with the atmosphere, 3Heexc = 3He
due
to excess air [29] and 3Heterr = terrigenic 3He
(nucleogenic 3He plus mantle 3He). In this
equation,
only 3Hetot and 3Heeq are
determined
through measurements. 3Heexc and 3Heterr
(if present in the sample) have to be determined by using measurements
of 4He and Ne (3Heterr). The total 4He
concentration measured in a groundwater sample can be written as:
![]() (3)
(3)
where 4Hetot = total measured 4He concentration in the groundwater sample, 4Heeq = 4He concentration in solubility equilibrium with the atmosphere, 4Heexc = 4He concentration caused by excess air, and 4Heterr = terrigenic 4He concentration (radiogenic 4He plus mantle 4He). If no terrigenic helium is contained in the groundwater sample, 3Hetrit can be calculated by using equation (4):
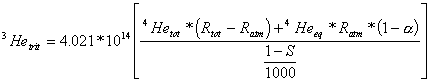 (4)
(4)
where Rtot = measured 3He/4He ratio of the sample, Ratm = 3He/4He ratio of air (1.384*10-6), a = solubility isotope effect (ca. 0.983)), and S = salinity in ‰. The conversion factor for 3He from cm3STPg-1 to TU is 4.021*1014/[(1-S)/1000)] TU/cm3STPg-1.
For separation of terrigenic helium, we have to use neon measurements. Assuming that neon has no sources other than the atmosphere, and that (Ne/4He)exc = (Ne/4He)air, we can calculate the terrigenic 4He component to be:
![]() (5)
(5)
where Netot = measured neon concentration in the sample, 4Heatm/Neatm = atmospheric He/Ne ratio (0.288), and (Netot - Neeq) = Neexc = neon concentration originating from excess air.
3Heterr is calculated by multiplying 4Heterr calculated from equation (5) by the 3He/4He ratio of the terrigenic helium component. This component can be radiogenic helium, mantle helium, or a mixture of these two components. The most practical approach to determining Rterr is to measure it in groundwater samples from the same aquifer that are free of tritium. If there is no tritium-free groundwater in the studied aquifer, an estimate of Rterr can be obtained in most cases by plotting 3He versus 4He. Such a plot typically provides fairly good clues with respect to the origin of the terrigenic helium.
The atmospheric 3He component (3Heatm) can be written as:
![]() (6)
(6)
Finally, 3Hetrit is obtained from equation (7) [e.g., 2]:
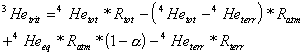 (7)
(7)
3. Non-linearity of the tritium/3He age
The tritium/3He age formally calculated from equation (1) is an apparent age. It is independent of the initial tritium concentration of the water sample which is one of the advantages of the method because it eliminates the necessity to establish the exact time- dependent tritium delivery to the aquifer. A potential problem for the quantitative interpretation of the tritium/3He age is the fact that it is affected by mixing and dispersion. Due to this effect, the tritium/3He age is typically biased toward the age of the water component with the higher tritium concentration. Therefore, for quantitative studies, mixing has either to be ruled out as a major factor influencing the flow regime or it has to be accounted for in the data evaluation.
Significant differences between the apparent tritium/3He ages and the true water ages typically occur near the location of the bomb peak. The reason for this observation is due to the high tritium and 3He concentration gradients near the bomb peak and the related increased transport of both tracers by dispersive processes. This effect also determines the useful time period over which tritium/3He dating is typically applicable. In many aquifers, the tritium/3He balance below the bomb tritium peak is dominated by downward dispersion of bomb tritium and 3Hetrit resulting from its radioactive decay. Therefore, in such cases, the tritium/3He age distribution is constant over a depth range of a few to several ten meters below the location of the bomb peak. However, in aquifers with very low dispersion rates, meaningful tritium/3He ages can be obtained for periods preceding the bomb peak.
4. Confinement of tritiogenic 3He
Because 3He is a gas, the application of the tritium/3He method to dating of groundwater depends strongly on the degree of confinement of 3Hetrit in the groundwater. The confinement of 3Hetrit is mainly determined by the ratio of advection to dispersion in water parcels moving away from the water table. It has been shown that for aquifers with reasonably high recharge rates (and related vertical flow velocities near the water table), loss of 3Hetrit is smaller than about 10 to 20%.